BDNF Methylation and Suicidal Ideation in Patients with Acute Coronary Syndrome
Article information
Abstract
Objective
Patients with acute coronary syndrome (ACS) are at an increased risk of suicide. It is well known that epigenetic mechanisms may explain the pathophysiology of suicidal behavior including suicidal ideation (SI), but no study has explored these mechanisms in ACS populations.
Methods
In total, 969 patients were initially recruited within 2 weeks of the acute coronary event and, 711 patients were successfully followed up 1 year after ACS. SI was evaluated using the relevant items on the Montgomery-Åsberg Depression Rating Scale and covariates potentially affecting SI were estimated.
Results
Brain-derived neurotrophic factor (BDNF) hypermethylation was associated with SI in both the acute and chronic phases of ACS, although the association was not statistically significant in the acute phase after applying Bonferroni’s correction.
Conclusion
These results suggested that BDNF hypermethylation may have played a role in an epigenetic predisposition for SI in ACS patients, particularly during the chronic phase.
INTRODUCTION
Acute coronary syndrome (ACS) is strongly related to a higher risk of suicide [1], and understanding the pathophysiology of suicide in ACS is important. Several biological mechanisms have been proposed to explain the pathophysiology of suicide [2]. Brain-derived neurotrophic factor (BDNF) affects both neuroplasticity and neurotransmission and has been widely investigated in the context of suicide. In several studies, reduced levels of BDNF, or alterations in the BDNF allele, have been associated with suicidality [3,4]. Additionally, BDNF may play an important role in atherosclerosis and the revascularization of ischemic tissue, with studies finding reduced BDNF levels in ACS patients [5]. Together, these associations suggest that BDNF could affect suicidality in ACS patients.
Epigenetic mechanisms regulate gene expression in response to environmental stimuli without changing nucleotide sequences. These mechanisms are thought to be capable of explaining complex phenomena, including suicidality [6]. Epigenetic modifications of BDNF expression may provide an alternative explanation for predictors of suicide in ACS patients, as these modifications reflect interactions between BDNF gene expression and environmental stressors, such as the ACS event. Methylation of the BDNF gene has been investigated in relation to suicidal behavior in general depression [7,8], because BDNF hypermethylation was associated with reduced BDNF expression [8]. However, there have been no studies evaluating the association between BDNF expression and suicidal behavior in ACS patients.
Suicidal behavior includes completed suicide, attempted suicide, and SI. SI is known to be crucial predictor of future suicide attempts and these thoughts themselves can cause a considerable burden [9]. Therefore, this study focuses on SI in ACS patients. Using the data from an ACS cohort, this study aimed to investigate the longitudinal association between SI and BDNF methylation status.
METHODS
This study was conducted as part of a prospective study of ACS patients, named the Korean DEPression in ACS (KDEPACS) study, which included a nested double-blind, randomized placebo-controlled trial: the Escitalopram for DEPression in ACS (EsDEPACS) study. The recruitment process for both studies has been described in a previous publication [10] and is reiterated in Supplementary Figure 1 (in the onlineonly Data Supplement). Written informed consents and approval by the Chonnam National University Hospital Institutional Review Board were obtained.
ACS patients recently admitted to the cardiology department were approached to participate. Those who consented to both participation and blood sampling (n=969) comprised the acute phase sample. All acute phase participants were reapproached at 1 year to assess SI status during the chronic post-ACS phase. Of 969 patients, 711 (73%) were successfully recruited after 1 year. There were no significant differences in characteristics between patients lost to follow-up and chronicphase participants, apart from an older mean age and higher Killip class (p<0.05).
SI was evaluated using the suicidal thoughts item of the Montgomery-Åsberg Depression Rating Scale (MADRS-ST). SI was evaluated within 2 weeks of the index acute coronary event, and at the 1-year follow-up [11]. The presence of SI was determined by a score of 2 (fleeting suicidal thoughts) or more, as described in previous studies [12]. Depression, sociodemographic, and cardiovascular characteristics potentially associated with SI in ACS patients [13] were investigated as covariates within 2 weeks of the index event, as described in Supplementary Table 1 (in the online-only Data Supplement).
BDNF methylation status was determined using the PSQ 96M Pyrosequencing System (Biotage AB, Uppsala, Sweden) and Pyro Q-CpG software (ver. 1.0.9; Biotage AB) with DNA taken from leukocytes. Methylation was assessed in a cytosine-guanine (CpG)-rich area lying between -612 and -463 relative to the transcriptional start of exon VII that included nine CpG sites (Supplementary Figure 2 in the online-only Data Supplement). This region has been reported to correspond to an analogous region in rat BDNF that is differentially methylation and associated with BDNF messenger RNA expression [14] and because it has been investigated in previous psychiatric studies [15,16]. CpG sites 2, 4, and 6 were excluded from the analysis as these sites showed 100% methylation in all participants. Thus, the methylation percentages at the six individual CpG sites, and the average value of all sites, were used in the analysis.
Statistical analysis
Demographic and clinical variables were compared according to SI status in both the acute and chronic ACS phases using t-tests or χ2 tests. Variables significantly associated with SI (p<0.05) were included as covariates in further adjusted regression models. Analyses of the association between BDNF methylation at individual CpG sites and SI, in both the acute and chronic disease phases, were evaluated using t-tests. The independent associations between the SI and methylation status were calculated using logistic regression models after adjusting for covariates. Treatment with antidepressants was also included as a covariate in the follow-up analysis, in line with previous studies [13]. Bonferroni’s correction was used to maintain an overall type 1 error rate of 0.05 in the context of seven comparisons (six CpG sites and the average value of all sites). A two-sided p-value of 0.007 (0.05/7) was taken to indicate statistical significance. All analyses were conducted using SPSS software (ver. 21.0; IBM Corp., Armonk, NY, USA).
RESULTS
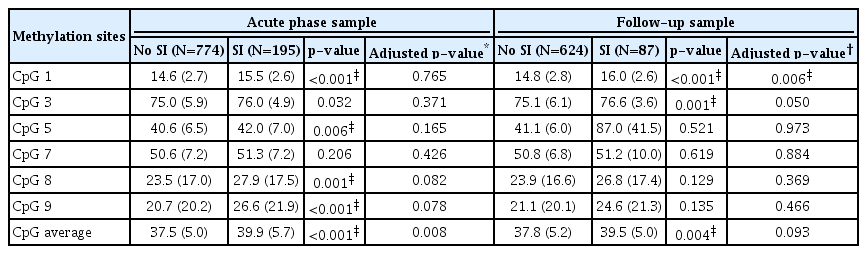
The BDNF methylation percentages of individual CpG sites, and the average value of all sites, were compared according to SI status at 2 weeks and 1 year after ACS (Table 1). Hypermethylation at five individual CpG sites (excluding CpG site 7), and the average value, were significantly associated with SI within 2 weeks of the acute coronary event. Hypermethylation of CpG sites 1 and 3, and the average value, were significantly associated with SI at 1 year following ACS. After Bonferroni correction, all acute-phase associations, except between the hypermethylation of CpG site 3 and SI, remained statistically significant. All chronic-phase associations remained statistically significant after Bonferroni correction.
The association between BDNF methylation and SI after covariate adjustment is presented in Table 1. The statistical significance of the association between hypermethylation of CpG site 9, and the average value, with SI within 2 weeks after ACS was lost after applying the Bonferroni correction (p=0.0078–0.008). However, the significant association between hypermethylation of CpG site 1 and SI at 1 year after ACS remained after correction.
DISCUSSION
This was the first study to explore longitudinal epigenetic vulnerability to SI in an ACS patient population. The data presented herein suggest that BDNF hypermethylation may predispose ACS patients to SI during both the acute and chronic phase, although the acute-phase association was not statistically significant after applying Bonferroni’s correction.
BDNF is important for neuroplasticity and neurotransmission, and hypermethylation has been associated with downregulation of BDNF expression [8]. The BDNF hypermethylation seen in this study may have resulted in decreased BDNF expression, impairing neural maintenance and neural plasticity. In turn, this may have led to a reduced ability to adapt to stressful situations and an increased risk of SI in ACS patients. Additionally, BDNF has been shown to directly interact with serotonin transportation in humans [17]. A hypermethylationinduced reduction in BDNF expression was associated with a reduction in the function of serotonin. Abnormal levels of serotonin are well-known to mediate suicide risk [2]. Further studies investigating the exact mechanisms by which BDNF hypermethylation is associated with SI are needed, including direct measurement of BDNF expression levels.
In this study, BDNF hypermethylation was associated with SI at 1 year after the ACS event, but not within 2 weeks after ACS. These findings reflect the fact that SI in ACS patients has a number of possible etiologies, where these vary according to the time after ACS diagnosis. In the acute phase, overwhelmed physiological and psychological responses, including heightened inflammatory or stress responses, may drive SI. However, during the chronic phase, BDNF hypermethylation may take on a more important role. This hypothesis is supported by several previous publications that outline the determinants of SI according to the time elapsed after the ACS event [13].
This is the first prospective investigation to evaluate epigenetic influences on SI in patients following ACS. Epigenetic modification is a crucial transducer of environmental influence [6]; thus, epigenetic factors may predict complex phenomena such as suicidality. This study is consistent with previous publications reporting that BDNF hypermethylation was associated with suicide in the postmortem brain [7], as well as with SI in patients with depression and breast cancer [8,18]. Although methylation status might differ by organ system, expression or methylation of BDNF in the central nervous system and blood stream are thought to be correlated due to the ability of BDNF to cross the blood-brain barrier [19]. Therefore, our findings suggest that BDNF hypermethylation in the peripheral blood may be a biological marker of SI, particularly in ACS patients. There were several additional strengths and limitations to the current study that should be considered; these are further outlined in the Supplementary Materials (in the online-only Data Supplement).
In conclusion, the data presented herein show that BDNF hypermethylation measured at acute phase of ACS predicted SI at 1 year later. BDNF methylation status could be a biomarker for suicidality in ACS, although the finding should be replicated. Future studies are needed to examine whether new drug that modulates methylation status may contribute to decrease SI in ACS patients, since DNA methylation is known to be reversible by pharmacological agents. We believe that our study serve the basis for epigenetic mechanisms in the etiology of SI in ACS.
Supplementary Materials
The online-only Data Supplement is available with this article at https://doi.org/10.30773/pi.2018.09.20.
Acknowledgements
This study was supported by a grant (BCRI18018) Chonnam National University Hospital Biomedical Research Institute and by a grant of the National Research Foundation of Korea Grant (NRF-2017M3A9E8023015).