 |
 |
- Search
| Psychiatry Investig > Volume 15(9); 2018 > Article |
|
Abstract
The concept of cognitive frailty has recently been proposed by an International Consensus Group as the presence of physical frailty and cognitive impairment [defined using the Clinical Dementia Ratings (CDR)=0.5], without concurrent dementia. However, CDR is difficult to implement and not often available in epidemiologic studies or busy clinical settings, and an alternative to CDR is required. We suggest an alternative definition of cognitive frailty as: 1) physical frailty, 2) more than 1.5 standard deviation below the mean for age-, gender-, and education-adjusted norms on any cognitive function test (e.g., the Montreal Cognitive assessment test, the Alzheimer’s disease assessment scale-cognitive subscale, verbal learning test, Digit Span, Boston Naming Test, Trail Making Test, and Frontal Assessment Battery), and 3) no dependency in instrumental activities of daily living. The redefined criteria for cognitive frailty would be more feasible to implement and thus more applicable in epidemiologic studies and busy clinical settings.
With increasing life expectancy, frailty and cognitive impairment are being recognized as major threats to healthy aging and quality of life. Frailty in late life predisposes the individual to increased vulnerability to stressors, elevating the risk of disability, institutionalization, and mortality [1]. Physical frailty is quite prevalent in older adults, with a recent systematic review reporting 9.9% of community-dwelling adults aged 65 years and older to have the condition [2]. With age-related cognitive decline, cognitive impairment that does not reach the threshold of dementia is commonly observed in older people, with an increased risk for progression to dementia, contributing to increased disability and healthcare costs [3].
Physical frailty and cognitive impairment often co-occur, with the two conditions being closely interrelated. About 20% of physically frail individuals living in the community are said to be cognitively impaired [4]. Several cohort studies have reported that physical frailty predicts the onset of cognitive decline and incident dementia [5-7]. Cognitive impairment has also been observed to predict physical frailty [8]. Moreover, physical frailty and cognitive impairment appear to reinforce each other, resulting in detrimental outcomes. Frailty when combined with mild cognitive impairment or dementia elevates the risk of poor outcomes [9].
Based on the results of these studies, a concept of ‚Äúcognitive frailty‚ÄĚ has been recently proposed by the International Consensus Group on Cognitive Frailty, organized by the International Academy on Nutrition and Aging (IANA) and the International Association of Gerontology and Geriatrics (IAGG) in France [8]. According to the International Consensus Group, cognitive frailty is defined as the 1) presence of physical frailty and cognitive impairment [Clinical Dementia Rating (CDR)=0.5] and 2) exclusion of concurrent dementia (Figure 1) [10].
The CDR is widely accepted in the clinical setting as a reliable and valid tool for assessing the severity of dementia [11]. However, the CDR is often not available or difficult to implement in epidemiologic studies or busy clinical settings. It is important to note that the CDR is a clinical protocol composed of semi-structured interviews where a clinician or neuropsychologist obtains information from the patient and proxy respondent, and then rates the cognitive performance of the patient in six domains: memory, orientation, judgment and problem solving, community affairs, home and hobbies, and personal care. Furthermore, epidemiologic field surveys sometimes entail recognizing and excluding possible dementia patients in the absence of dementia specialists or neuropsychologists. Therefore, the CDR may not be appropriate for use in epidemiologic studies because of its complexity in measurement and difficulty to implement in the field settings. Thus, we suggest alternative criteria to CDR 0.5 and exclusion of dementia for diagnosing cognitive frailty.
Based on the definition by the International Consensus Group of IANA and IAGG, a CDR score 0.5 and the exclusion of concurrent dementia correspond to the concept of mild cognitive impairment (MCI). Many investigators have used a CDR score of 0.5 to define MCI. However, a CDR score of 0.5 is not equivalent to the diagnosis of MCI [12]. In one study, 39.7% of people with a CDR score of 0.5 were found to be demented [13]. Therefore, the diagnosis of MCI and the exclusion of dementia cannot be determined with the CDR score alone.
How can we devise an alternative to the CDR score of 0.5 and the exclusion of concurrent dementia? Petersen’s original criteria of MCI used neuropsychological (NP) test performance and operationalized MCI as a score of more than 1.5 standard deviations below that of age-appropriate norms on a measure of episodic memory with performance within the normal range on nonmemory tests [14]. Using a similar approach, the International Working Group on MCI has adapted Petersen’s criteria to take into account differing patterns of cognitive test performance and allowed either memory or nonmemory impairment and single cognitive domain or multiple impaired domains [15].
As to the cognitive assessment for cognitive frailty, the International Consensus Group on Cognitive Frailty by IANA and IAGG have already suggested comprehensive cognitive assessment exploring memory performance as well as other cognitive functions (i.e., executive functions). The panel have also suggested several cognitive tests such as speed of processing test, the Montreal Cognitive assessment (MoCA) test, the Mini Mental state Examination (MMSE) and the Alzheimer’s disease assessment scale-cognitive subscale (ADAS-Cog) [10].
However, the MMSE had low sensitivity (66.01%) for MCI by traditionally accepted cut-off points set at 26/27 [16]. On the other hand, for MoCA test, the best cut-off point is 24/25 with sensitivity of 80.48% and specificity of 81.19% [17].
Among cognitive abilities, the frontal lobe function is important and needs to be assessed for diagnosis of cognitive frailty. This is because cognitively frail individuals might demonstrate significantly more impairments in executive functions than individuals with cognitive impairment but without physical frailty [18]. Executive functions primarily affected in the cognitive frailty were processing speed, selective attention, and mental flexibility, reflecting sub-cortico-frontal cognitive patterned impairments [18].
To diagnose cognitive frailty, we should exclude dementia as well. In 2011, a working group charged by the National Institute on Aging and the Alzheimer’s Association revised the diagnostic criteria for MCI [19] (Table 1). The criteria for dementia stipulate that cognitive impairment must be present in two or more domains and must interfere with the abilities to function in daily activities [19,20]. A key criterion for MCI is the absence of dementia. Thus, individuals have to experience cognitive impairment while keeping their functioning abilities intact. Therefore, the preservation of functional activities is essential to differentiating MCI from dementia [15,21].
Based on the above reviews, we propose the new criteria of cognitive frailty as follows (Figure 2).
1) physical frailty; 2) 1.5 SD below the mean for age-, gender-, and education-adjusted norms on any cognitive functioning test (e.g., the MoCA, the ADAS-Cog, verbal learning test, Digit Span, Boston Naming Test, Trail Making Test, and Frontal Assessment Battery); and 3) no dependency in instrumental activities of daily living (IADL); Among IADL items, financial capacity (managing money), telephone use, responsibility for medication, and keeping appointments should be included because these items are particularly more complex tasks requiring high cognitive demands [18].
In conclusion, we propose a new definition of cognitive frailty for wider use in epidemiologic studies and busy clinical settings. The revised criteria need to be tested to determine its value in accurately assessing cognitive frailty and predicting its outcomes.
ACKNOWLEDGEMENTS
This research was supported by a grant of the Korea Health Technology R&D Project through the Korean Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI15C3153).
Figure 1.
Definition of cognitive frailty by IANA/IAGG International Consensus Group. Cognitive frailty is defined as the presence of physical frailty and cognitive impairment (CDR=0.5) and the exclusion of concurrent dementia. CDR=0.5 includes mild cognitive impairment (MCI) and some dementia, but the exclusion of concurrent dementia limits the boundary to MCI. Cognitive frailty corresponds to the area indicated by the arrow.
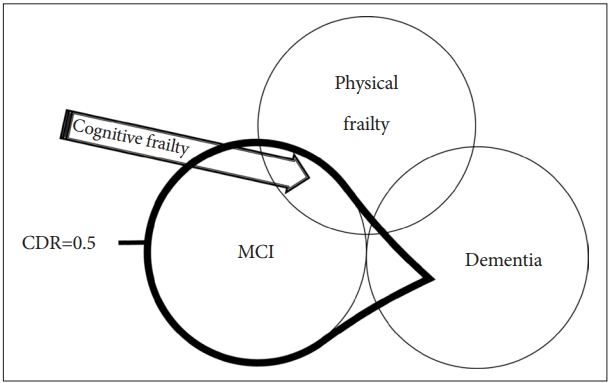
Figure 2.
Newly suggested definition of cognitive frailty for epidemiologic studies. Any cognitive function test < -1.5 SD of the age-, gender-, and education-adjusted norms mean MCI or Dementia. Dependency in IADL encompasses dementia. The newly suggested definition of cognitive frailty (arrow) is physical frailty and any cognitive function test < -1.5 SD of the age-, gender-, and education-adjusted norm and no dependency in IADL. MCI: mild cognitive impairment, IADL: instrumental activities of daily living.
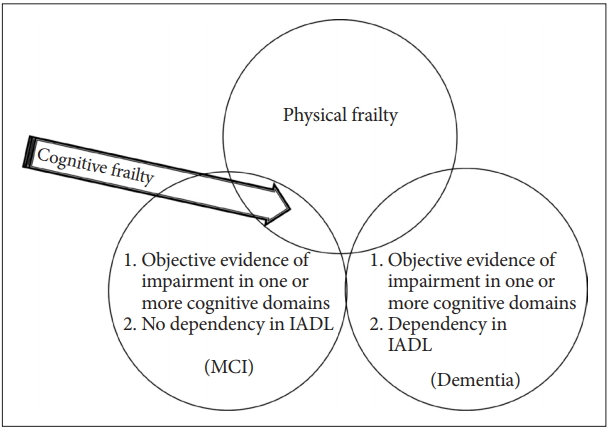
Table 1.
The revised criteria for mild cognitive impairment by the working group organized by the National Institute on Aging and the Alzheimer’s Association
REFERENCES
1. Morley JE, Vellas B, van Kan GA, Anker SD, Bauer JM, Bernabei R, et al. Frailty consensus: a call to action. J Am Med Dir Assoc 2013;14:392-397.



2. Collard RM, Boter H, Schoevers RA, Oude Voshaar RC. Prevalence of frailty in community‚Äźdwelling older persons: a systematic review. J Am Geriatr Soc 2012;60:1487-1492.


3. Plassman BL, Langa KM, Fisher GG, Heeringa SG, Weir DR, Ofstedal MB, et al. Prevalence of cognitive impairment without dementia in the United States. Ann Intern Med 2008;148:427-434.



4. √Āvila-Funes JA, Amieva H, Barberger-Gateau P, Le Goff M, Raoux N, Ritchie K, et al. Cognitive impairment improves the predictive validity of the phenotype of frailty for adverse health outcomes: the three-city study. J Am Geriatr Soc 2009;57:453-461.


5. Boyle PA, Buchman AS, Wilson RS, Leurgans SE, Bennett DA. Physical frailty is associated with incident mild cognitive impairment in community-based older persons. J Am Geriatr Soc 2010;58:248-255.



6. Buchman AS, Boyle PA, Wilson RS, Tang Y, Bennett DA. Frailty is associated with incident Alzheimer’s disease and cognitive decline in the elderly. Psychosom Med 2007;69:483-489.


7. Robertson DA, Savva GM, Kenny RA. Frailty and cognitive impairment--a review of the evidence and causal mechanisms. Ageing Res Rev 2013;12:840-851.


8. Raji MA, Al Snih S, Ostir GV, Markides KS, Ottenbacher KJ. Cognitive status and future risk of frailty in older Mexican Americans. J Gerontol A Biol Sci Med Sci 2010;65:1228-1234.



9. Song X, Mitnitski A, Rockwood K. Nontraditional risk factors combine to predict Alzheimer disease and dementia. Neurology 2011;77:227-234.



10. Kelaiditi E, Cesari M, Canevelli M, Van Kan GA, Ousset P, Gillette-Guyonnet S, et al. Cognitive frailty: rational and definition from an (IANA/IAGG) international consensus group. J Nutr Health Aging 2013;17:726-734.



11. Morris JC, Storandt M, Miller JP, McKeel DW, Price JL, Rubin EH, et al. Mild cognitive impairment represents early-stage Alzheimer disease. Arch Neurol 2001;58:397-405.


12. Kim JW, Lee DY, Seo EH, Sohn BK, Choe YM, Kim SG, et al. Improvement of screening accuracy of mini-mental state examination for mild cognitive impairment and non-Alzheimer’s disease dementia by supplementation of verbal fluency performance. Psychiatry Investig 2014;11:44-51.



13. Morris JC. Clinical dementia rating: a reliable and valid diagnostic and staging measure for dementia of the Alzheimer type. Int Psychogeriatr 1997;9(Suppl 1):173-176.


14. Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E. Mild cognitive impairment: clinical characterization and outcome. Arch Neurol 1999;56:303-308.


15. Winblad B, Palmer K, Kivipelto M, Jelic V, Fratiglioni L, Wahlund L, et al. Mild cognitive impairment-beyond controversies, towards a consensus: report of the International Working Group on Mild Cognitive Impairment. J Intern Med 2004;256:240-246.


16. Xu G, Meyer JS, Thornby J, Chowdhury M, Quach M. Screening for mild cognitive impairment (MCI) utilizing combined mini-mental-cognitive capacity examinations for identifying dementia prodromes. Int J Geriatr Psychiatry 2002;17:1027-1033.


17. Ciesielska N, Sokolowski R, Mazur E, Podhorecka M, Polak-Szabela A, Kedziora-Kornatowska K. Is the Montreal Cognitive Assessment (MoCA) test better suited than the Mini-Mental State Examination (MMSE) in mild cognitive impairment (MCI) detection among people aged over 60? Meta-analysis. Psychiatr Pol 2016;50:1039-1052.



18. Delrieu J, Andrieu S, Pahor M, Cantet C, Cesari M, Ousset P, et al. Neuropsychological profile of ‚Äúcognitive frailty‚ÄĚ subjects in MAPT Study. J Prev Alzheimers Dis 2016;3:151



19. Albert MS, DeKosky ST, Dickson D, Dubois B, Feldman HH, Fox NC, et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 2011;7:270-279.









