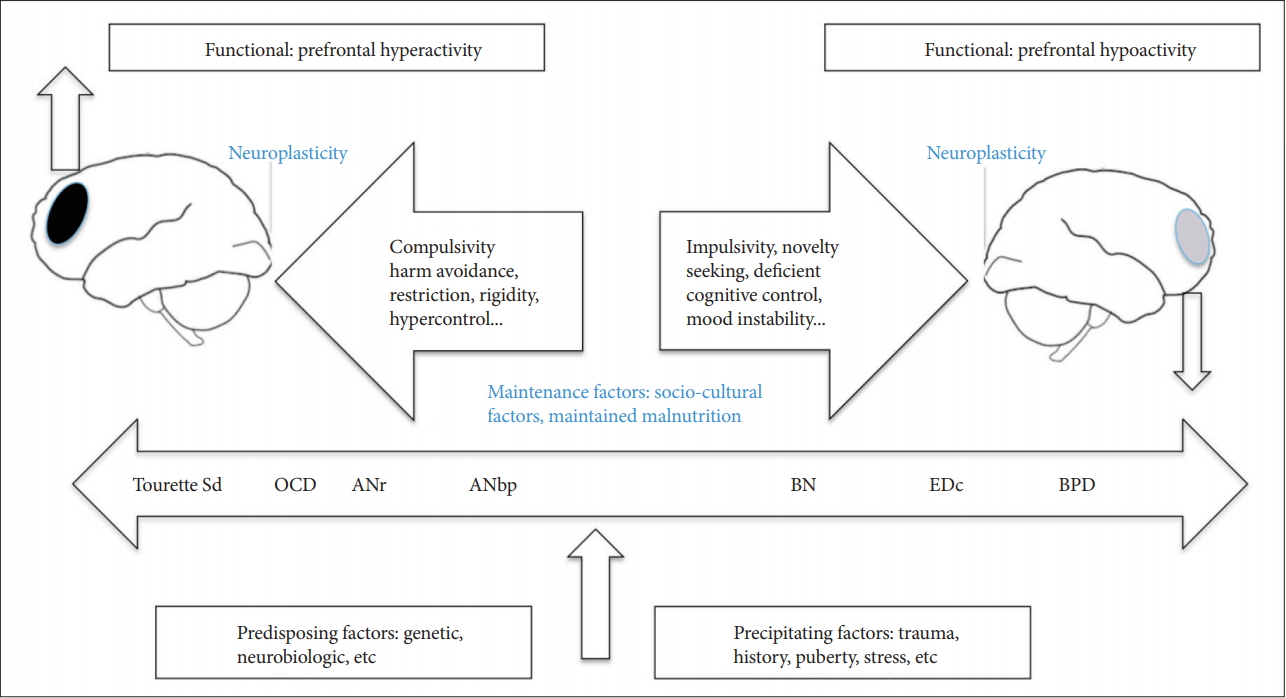
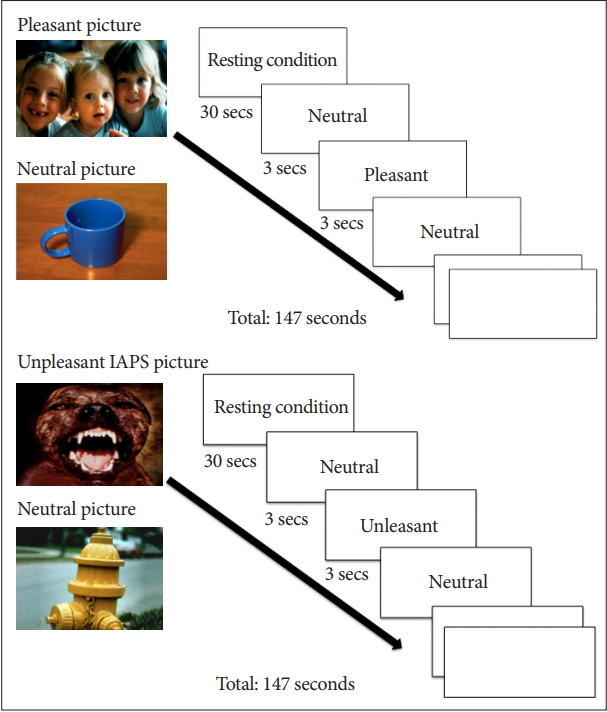
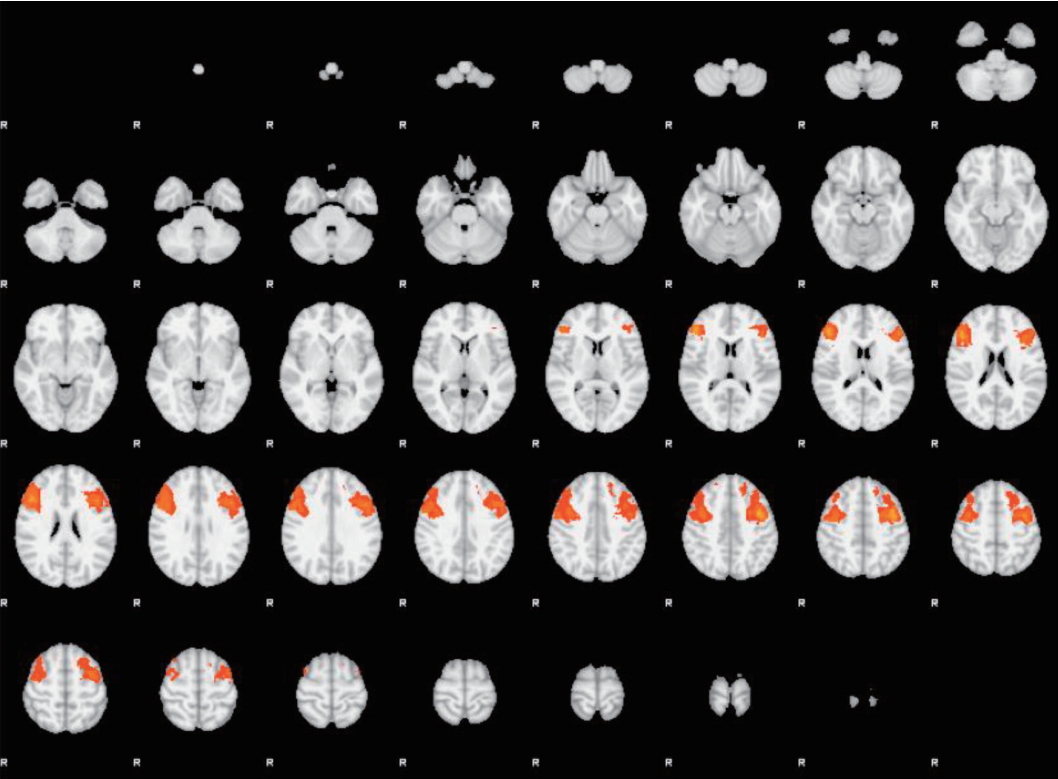
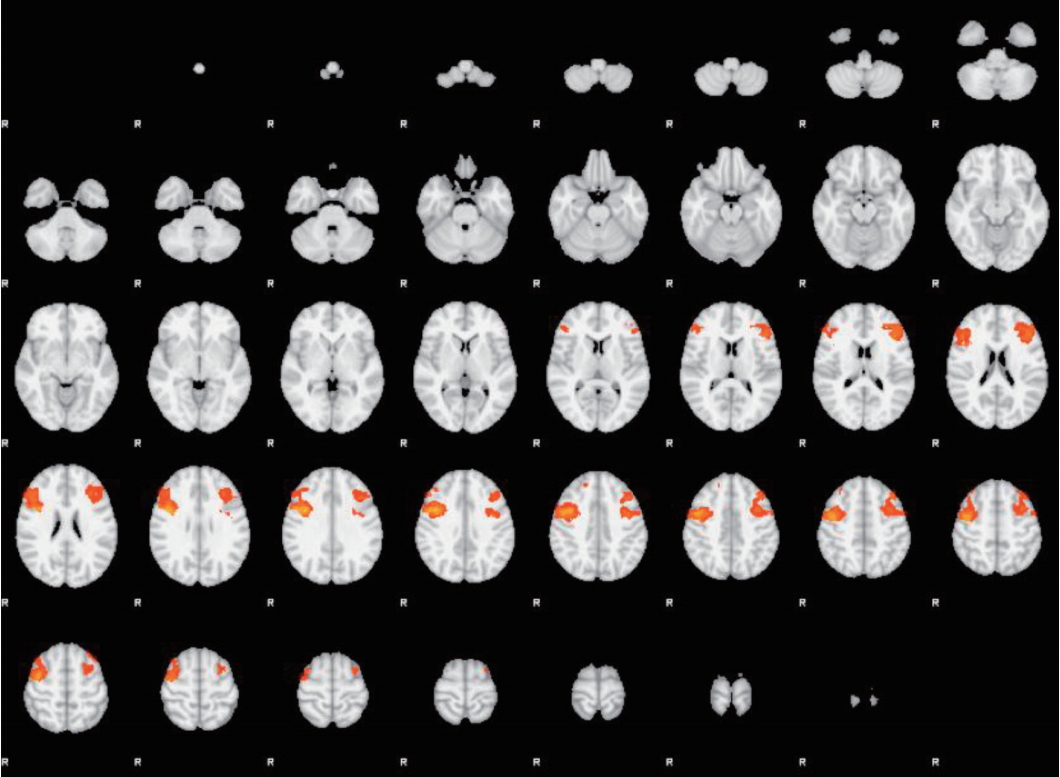
2. Oldershaw A, Hambrook D, Stahl D, Tchanturia K, Treasure J, Schmidt U. The socio-emotional processing stream in Anorexia Nervosa. Neurosci Biobehav Rev 2011;35:970-988.


3. Harrison A, Sullivan S, Tchanturia K, Treasure J. Emotional functioning in eating disorders: attentional bias, emotion recognition and emotion regulation. Psychol Med 2010;40:1887-1897.


4. Espina Eizaguirre A, Ortego Saenz de Cabezón A, Ochoa de Alda I, Joaristi Olariaga L, Juaniz M. Alexithymia and its relationships with anxiety and depression in eating disorders. Pers Individ Dif 2004;36:321-331.

5. Gilboa-schechtman E, Avnon ÃL, Zubery E, Jeczmien P. Emotional processing in eating disorders: specific impairment or general distress related deficiency? Depress Anxiety 2006;23:331-339.


7. Kelley AE. Ventral striatal control of appetitive motivation: role in ingestive behavior and reward-related learning. Neurosci Biobehav Rev 2004;27:765-776.


8. Kaye WH, Wierenga CE, Bailer UF, Simmons AN, Bischoff-Grethe A. Nothing tastes as good as skinny feels: the neurobiology of anorexia nervosa. Trends Neurosci 2013;36:110-120.


11. Meyer C, Waller G, Waters A. Emotional States and Bulimic Psychopathology. In: Hoek H, editor. Neurobiology in the Treatment of Eating Disorders. Chichester: Wiley, 1998, p. 271-289.
13. Waters A, Hill A, Waller G. Bulimics’ responses to food cravings: is binge-eating a product of hunger or emotional state? Behav Res Ther 2001;39:877-886.


15. Lacey JH, Evans CD. The Impulsivist: a multi-impulsive personality disorder. Addiction 1986;81:641-649.

16. Herzog DB, Keller MB, Lavori PW, Kenny GM, Sacks NR. The prevalence of personality disorders in 210 women with eating disorders. J Clin Psychiatry 1992;53:147-152.

17. Skodol AE, Oldham JM, Hyler SE, Kellman HD, Doidge N, Davies M. Comorbidity of DSM-III-R eating disorders and personality disorders. Int J Eat Disord 1993;14:403-416.


18. Koepp W, Schildbach S, Schmager C, Rohner R. Borderline diagnosis and substance abuse in female patients with eating disorders. Int J Eat Disord 1993;14:107-110.


20. Hollander E, Evers M. Review of obsessive-compulsive spectrum disorders: what do we know? where are we going? Clin Neuropsychiatry J Treat Eval 2004;1:32-51.
21. Skodol AE, Oldham JM. Phenomenology, Differential Diagnosis and Comorbidity of the Impulsive-Compulsive Spectrum Disorders. In: Oldham JM, Hollander E, editor. Impulsivity and Compulsivity. Washington DC, London: American Psychiatric Press, Inc, 1996, p. 1-26.
23. Chamberlain SR, Blackwell AD, Fineberg NA, Robbins TW, Sahakian BJ. The neuropsychology of obsessive compulsive disorder: the importance of failures in cognitive and behavioural inhibition as candidate endophenotypic markers. Neurosci Biobehav Rev 2005;29:399-419.


24. Stein DJ, Lochner C. Obsessive-compulsive spectrum disorders: a multidimensional approach. Psychiatr Clin North Am 2006;29:343-351.


26. Baxter LR. Neuroimaging studies of obsessive compulsive disorder. Psychiatr Clin North Am 1992;15:871-884.


27. Baxter L, Saxena S, Brody A, Ackermann R, Colgan M, Schwartz J, et al. Brain mediation of obsessive-compulsive disorder symptoms: evidence from functional brain imaging studies in the human and nonhuman primate. Semin Clin Neuropsychiatry 1996;1:32-47.

28. Frank GK, Bailer UF, Henry S, Wagner A, Kaye WH. Neuroimaging studies in eating disorders. CNS Spectr 2004;9:539-548.


30. Ellison ZR, Foong J. Neuroimaging in eating disorders. In: Hoek HW, Treasure J, Melanie A, editor. Neurobiology in the Treatment of Eating Disorders. West Sussex: John Wiley & Sons Ltd, 1998, p. 255
31. Wagner A, Matthias R, Dieter B, Schmidt M. Neuronal activity changes and body image distortion in anorexia nervosa. Neuroreport 2003;14:2193-2197.


32. Uher R, Murphy T, Friederich HC, Dalgleish T, Brammer MJ, Giampietro V, et al. Functional neuroanatomy of body shape perception in healthy and eating-disordered women. Biol Psychiatry 2005;58:990-997.


33. Fladung AK, Grön G, Grammer K, Herrnberger B, Schilly E, Grasteit S, et al. A neural signature of anorexia nervosa in the ventral striatal reward system. Am J Psychiatry 2010;167:206-212.


34. Miyake Y, Okamoto Y, Onoda K, Shirao N, Okamoto Y, Otagaki Y, et al. Neural processing of negative word stimuli concerning body image in patients with eating disorders: an fMRI study. Neuroimage 2010;50:1333-1339.


36. Seeger G, Braus DF, Ruf M, Goldberger U, Schmidt MH. Body image distortion reveals amygdala activation in patients with anorexia nervosa-a functional magnetic resonance imaging study. Neurosci Lett 2002;326:25-28.


37. Sachdev P, Mondraty N, Wen W, Gulliford K. Brains of anorexia nervosa patients process self-images differently from non-self-images: an fMRI study. Neuropsychologia 2008;46:2161-2168.


38. Gizewski E, Rosenberger C, de Greiff A, Moll A, Senf W, Wanke I, et al. Influence of satiety and subjective valence rating on cerebral activation patterns in response to visual stimulation with high-calorie stimuli among restrictive anorectic and control women. Neuropsychobiology 2010;62:182-192.


39. Esposito R, Cieri F, di Giannantonio M, Tartaro A. The role of body image and self-perception in anorexia nervosa: the neuroimaging perspective. J Neuropsychol 2018;12:41-52.


40. Spangler DL, Allen MD. An fMRI investigation of emotional processing of body shape in bulimia nervosa. Int J Eat Disord 2012;45:17-25.


41. Lang PJ, Bradley MM, Cuthbert BN. International Affective Pictures System (IAPS): Technical Manual and Affective Ratings. Gainesville, FL: NIMH Cent Study Emot Attention, University of Florida; 2001.
42. Tapajóz P, de Sampaio F, Soneira S, Aulicino A, Harris P, Allegri R. Emotional reactivity to social stimuli in patients with eating disorders. Psychiatry Res 2015;229:887-894.


44. Takahashi H, Koeda M, Oda K, Matsuda T, Matsushima E, Matsuura M, et al. An fMRI study of differential neural response to affective pictures in schizophrenia. Neuroimage 2004;22:1247-1254.


45. Britton JC, Phan KL, Taylor SF, Welsh RC, Berridge KC, Liberzon I. Neural correlates of social and nonsocial emotions: An fMRI study. Neuroimage 2006;31:397-409.


46. Grimm S, Schmidt CF, Bermpohl F, Heinzel A, Dahlem Y, Wyss M, et al. Segregated neural representation of distinct emotion dimensions in the prefrontal cortex-an fMRI study. Neuroimage 2006;30:325-340.


47. Berlin HA, Rolls ET, Iversen SD. Borderline personality disorder, impulsivity, and the orbitofrontal cortex. Am J Psychiatry 2005;162:2360-2373.


48. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-IV). 4th Ed. Washington, DC: American Psychiatry Press; 2000.
49. Spitzer RL, Williams JB, Gibbon M, First MB. The Structured Clinical Interview for DSM-III-R (SCID). I: History, rationale, and description. Arch Gen Psychiatry 1992;49:624-629.


50. Patton JH, Stanford MS, Barratt ES. Factor structure of the Barratt impulsiveness scale. J Clin Psychol 1995;51:768-774.


52. Henderson M, Freeman CP. A self-rating scale for bulimia: The “BITE. Br J Psychiatry 1987;150:18-24.


53. Rivas T, Bersabé R, Jiménez M. Reliability and validity of Bulimic Investigatory Test, Edinburgh (BITE)-Behavioral Psychology / PsicologÍa Conductual. Behav Psychol 2004;12:447-461.
54. Jayaro C, de la Vega I, DÍaz-Marsá M, Montes A, Carrasco JL. The use of the International Affective Picture System for the study of affective dysregulation in mental disorders. Actas españolas Psiquiatr 2008;36:177-182.
55. Desikan RS, Ségonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 2006;31:968-980.


56. Joos AA, Saum B, van Elst LT, Perlov E, Glauche V, Hartmann A, et al. Amygdala hyperreactivity in restrictive anorexia nervosa. Psychiatry Res 2011;191:189-195.


57. Adolphs R. Neural systems for recognizing emotion. Curr Opin Neurobiol 2002;12:169-177.


58. Cancelliere AE, Kertesz A. Lesion localization in acquired deficits of emotional expression and comprehension. Brain Cogn 1990;13:133-147.


59. Davidson R, Irwin W. The functional neuroanatomy of emotion and affective style. Trends Cogn Sci 1999;3:11-21.


60. Ochsner KN, Gross JJ. The cognitive control of emotion. Trends Cogn Sci 2005;9:242-249.


61. Vuilleumier P. How brains beware: neural mechanisms of emotional attention. Trends Cogn Sci 2005;9:585-594.


63. Hariri AR, Bookheimer SY, Mazziotta JC. Modulating emotional responses: effects of a neocortical network on the limbic system. Neuroreport 2000;11:43-48.


64. Lieberman MD, Eisenberger NI, Crockett MJ, Tom SM, Pfeifer JH, Way BM. Putting feelings into words: affect labeling disrupts amygdala activity in response to affective stimuli. Psychol Sci 2007;18:421-428.


67. Geller J, Cockell SJ, Hewitt PL, Goldner EM, Flett GL. Inhibited expression of negative emotions and interpersonal orientation in anorexia nervosa. Int J Eat Disord 2000;28:8-19.


69. Van Kuyck K, Gérard N, Van Laere K, Casteels C, Pieters G, Gabriëls L, et al. Towards a neurocircuitry in anorexia nervosa: evidence from functional neuroimaging studies. J Psychiatr Res 2009;43:1133-1145.


70. Zastrow A, Kaiser S, Stippich C, Walther S, Herzog W, Tchanturia K, et al. Neural correlates of impaired cognitive-behavioral flexibility in anorexia nervosa. Am J Psychiatry 2009;166:608-616.


71. Adan W, Kaye WH. Behavioral Neurobiology of Eating Disorders. Berlin: Springer; 2011.
72. Klump KL, Strober M, Bulik CM, Thornton L, Johnson C, Devlin B, et al. Personality characteristics of women before and after recovery from an eating disorder. Psychol Med 2004;34:1407-1418.


73. Uher R, Murphy T, Brammer MJ, Dalgleish T, Phillips ML, Ng VW, et al. Medial prefrontal cortex activity associated with symptom provocation in eating disorders. Am J Psychiatry 2004;161:1238-1246.


74. Guido KWF, Bailer UF, Meltzer CC, Price JC, Mathis CA, Wagner A, et al. Regional cerebral blood flow after recovery from anorexia or bulimia nervosa. Int J Eat Disord 2007;40:488-492.


79. Grillner S, Hellgren J, Ménard A, Saitoh K, Wikström MA. Mechanisms for selection of basic motor programs--roles for the striatum and pallidum. Trends Neurosci 2005;28:364-370.


80. Bohon C, Stice E. Reward abnormalities among women with full and subthreshold bulimia nervosa: a functional magnetic resonance imaging study. Int J Eat Disord 2011;44:585-595.


82. Uher R, Brammer MJ, Murphy T, Campbell IC, Ng VW, Williams SCR, et al. Recovery and chronicity in anorexia nervosa: brain activity associated with differential outcomes. Biol Psychiatry 2003;54:934-942.