Skin Diseases in Patients with Primary Psychiatric Disorders
Article information
Abstract
Objective
The few psychodermatological studies of primary psychiatric populations so far suggest that parasitic-infectious skin diseases are the most common dermatological comorbidity in more than 70% of psychiatric patients, which should be studied here in a large data bank outside dermatological treatment facilities.
Methods
In a descriptive-explorative and retrospective study, more than 17,000 patients with primary psychiatric disorders were examined to investigate dermatological comorbidities.
Results
The proportion of patients with primary mental disorders and additional dermatological disease was 1.24% (n=212). Here, psoriasis (35.4%) and atopic dermatitis (22.6%) were the most frequent dermatological diseases among these 212 patients. Infectious-parasitic skin diseases were present in 13.2% of comorbid patients. The most common mental disorder was a depressive illness, seen in 42.5% (n=90) of patients.
Conclusion
Our results confirmed the frequent association of depression with psoriasis and atopic dermatitis, indicating the need for the early detection and treatment of such comorbid patients. In contrast, psychiatric inpatients do not appear to suffer from predominantly infectious-parasitic dermatoses.
INTRODUCTION
Both the skin and the central nervous system (CNS) are derived from ectoderm during embryogenesis; their resulting anatomical-functional relationship has long been known. Nevertheless, many aspects of this partially obvious connection are not fully understood. In particular, the complexity of the structure and the meticulous functions of both the skin and the CNS, but also the multiple interactions between them, complicate the investigation of pathological reactions and phenomena. This is reflected even in the rather poor state of studies with regard to the comorbid occurrence of mental disorders and skin diseases, although there are a number of publications which confirm an association between dermatoses and mental illness. In a multicentre study in 13 European countries, a close link between depression, anxiety disorders, and skin diseases was demonstrated on the basis of 3,635 dermatologically ill participants and a control group of 1,359 healthy individuals [1]. In this study, depressive disorders (10.1% vs. 4.3%) and anxiety disorders (17.2% vs. 11.1%) were the most comorbid mental diseases in patients with primary psoriasis, atopic dermatitis, hands’ eczema and ulcus cruris [1].
Such investigations, alongside most of the published studies, were usually performed in dermatological or psychodermatological-psychosomatic treatment facilities [2-7], but rarely in psychiatric hospitals [8,9]. However, the isolated findings from studies conducted in primary psychiatric populations suggest that infectious-parasitic dermatoses are the most common skin diseases in over 70% of patients with mental disorders. Researchers have discussed a possible explanation for this high comorbidity rate: the overall reduced physical condition of many mentally ill patients, which is associated with a reduced immune defense and thus increased susceptibility to infections of the skin [8,9].
Even though, in spite of this understandable explanation, these findings suggest that most patients with mental illnesses (and especially patients with schizophrenia) are incapable of performing their day-to-day hygiene and health care responsibilities, this impression should also be subjected to a critical evaluation due to aspects of stigmatization, which are widespread concerning psychiatric patients.
The aim of our descriptive-exploratory, the retrospective data-based study was, therefore, to further examine patients with a primary mental disorder in regard to dermatological comorbidities.
METHODS
First, in order to collect data, all psychiatric cases (ICD-10: F00–F99) were identified from a total of 5,071 patients hospitalized at the Department of Psychiatry (LWL-University Hospital Bochum) between 1984 and 2006 who also had a skin disease (ICD-10: L00–L99) according to the medical records. Here, the criterion of comorbidity met n=86 patients.
In addition, a second total sample of 12,014 patients was selected from the annual electronic records, according to § 21 KHEntG (Krankenhausentgeltgesetz), in the period between 2007 to 2015 under the inclusion criterion mentioned above (n=126). This second group consisted of patients who had been treated because of their mental illness either in the Department of Psychiatry (n=90) or in the Department of Psychosomatic Medicine (n=36) of the LWL-University Hospital Bochum.
In a further step, the population of all 212 comorbid patients resulting from the selection was assigned to eight different groups based on their additional dermatological diagnosis (Figure 1). This division was made on the basis of the psychodermatological disorder patterns frequently mentioned in the literature [10]. Subsequently, the patient-related socio-demographic, disease specific and clinical data were transferred to the SPSS file and subjected to further statistical analysis.

Flow diagram to identify patients groups being comorbid with psychiatric and dermatological disorders.
Apart from the presence of a mental disorder without a comorbid dermatological disease, no further exclusion criteria were defined. Special care was taken to ensure that two selected collectives did not consist of the cases (“Fall-IDs”), but of the once-allocated patient-IDs that corresponded to each hospital stay in order to avoid duplicates.
The study was approved by the local university ethics committee (Nr: 15-5347) of Ruhr University Bochum, Germany.
Statistical analysis
Further statistical analyses of the data were performed using IBM SPSS Statistics for Windows, Version 24.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics are given as mean values, standard deviation, and range. Additionally, statistical analyses were performed by appropriate parametric or nonparametric tests. Statistical significance was defined as p<0.05. A value of p<0.10 was regarded as a statistical tendency.
RESULTS
The proportion of patients with a primary psychiatric disorder and additional dermatological disease was 1.24% [212 of 17,085 patients; 116 females (54.7%) and 96 males (45.3%); patients had a mean age of 44.38±16.8 years].
About half of all comorbid patients (51.9%) had no permanent partner at the time of their inpatient treatment, 61 patients (28.8%) were married and 38 (17.9%) had a solid partnership.
In the majority of the comorbid patients (66%), we found an overall work impairment conditioned by unemployment (n=78) or retirement (n=62). Furthermore, 9.4% (n=20) of the patient population were students.
Prevalence of psychiatric morbidity
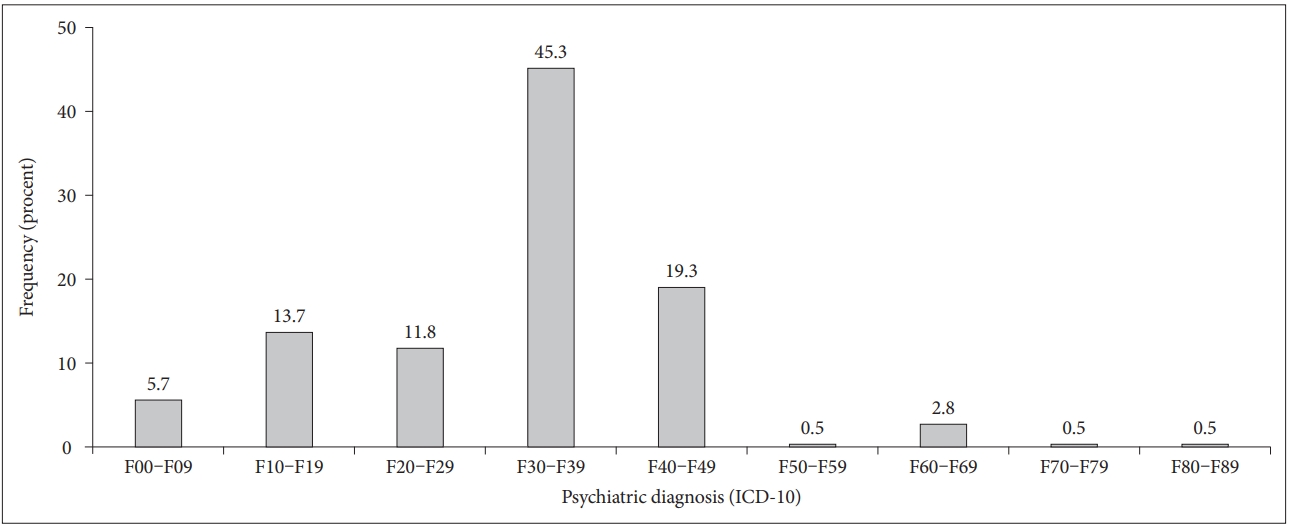
The primary psychiatric disorders in the studied group are shown in Figure 2. The most common psychiatric diagnoses (n=96, 45.3%) were mood disorders according to ICD-10: F30–F39. From these patients, 90 (42.5%) suffered from depression, more than half of whom (n=61, 63.54%) had a recurrent depressive disorder (F33.-).
The second largest group (19.3%, n=41) consisted of patients with neurotic, stress-related and somatoform disorders (ICD-10: F40–F48). Here, the majority of patients (n=21, 51.22%) showed reactions to severe stress and adjustment disorders (ICD-10: F43.-), followed by somatoform disorders (ICD-10: F45.-; n=9, 21.95%).
Moreover, 13.7% (n=29) of the group suffered from psychoactive drug-induced psychiatric disorders (ICD-10: F10–F19) and so was the third largest, where ethanol was the most common psychoactive substance (n=26, 89.7%).
Furthermore, 11.8% (n=25) of the comorbid patients had a disease from the spectrum of schizophrenia, schizotypal and delusional disorders (ICD-10: F20–F29).
The number of patients with an organic delusional disorder, including symptomatic mental disorders (ICD-10: F00–F09), was n=12 (5.7%). Finally, just six patients (2.8%) had a documented personality and behavioral disorder (ICD-10: F60–F69) (Figure 2).
Prevalence of dermatologic morbidity
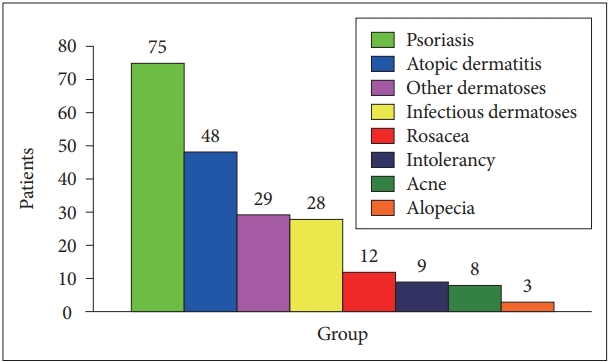
See Figure 3 regarding the distribution of common skin diseases among comorbid patients (n=212). The most frequent diagnostic groups were psoriasis (n=75, 35.4%) and atopic dermatitis (n=48, 22.6%).
Prevalence of comorbidity
The most frequent comorbid diagnoses were psoriasis with affective disorders (n=33, 15.7%) and atopic dermatitis with affective disorders (n=25, 11.8%). In detail, 29 of these psoriasis patients had depression and four suffered from a bipolar (manic-depressive) disorder. Of the atopic dermatitis patients, 23 had depression and two suffered from manic-depressive disorder.
Among the infectious-parasitic dermatoses, there was a depression in six patients, dementia in five patients, while four had schizophrenia and three experienced alcoholism. Table 1 illustrates the frequency of the comorbid infectious-parasitic skin diseases in more detail.
Comorbidity and somatic factors
It should be emphasized that in addition to the psychiatric and dermatological diseases, 30% (n=68) of our total comorbid collective also had cardiovascular diseases (especially arterial hypertension) and, moreover, diabetic metabolic states. Of the patients with infectious-parasitic skin diseases, more than half (53.6%) had an additional somatic disease.
DISCUSSION
In the present retrospective, naturalistic data-based study of more than 17,000 mentally ill inpatients, we found a 1.24% period prevalence for the comorbid occurrence of psychiatric disorders and skin diseases. Our findings contrast with the findings of previous studies, which found comorbidity rates between 10% and 60% [1,2,4,7]. However, it has to be taken into account that these prevalence rates were published almost exclusively in studies from dermatological clinics. On the other hand, so far only a few studies have been published from psychiatric or psychosomatic treatment facilities [8,9], but ultimately they do not allow a direct comparison of prevalence rates due to the existence of methodological differences.
Another possible explanation for the different prevalence could be that the patients with psychiatric illnesses and additional skin symptoms, to avoid social taboos and stigmatization, primarily seek dermatological treatment in specialized facilities [11], though it is generally assumed that 30% of primary dermatologically ill patients have an additional mental disorder [12,13]; this seems to be especially true for psoriasis and atopic dermatitis (neurodermatitis). Our results of prevalence rates of 35.4% of co-morbid psoriasis and 22.6% of atopic dermatitis in patients with a mental disorder are consistent with the findings of other studies [7,14-21].
The same also applies to our findings of the occurrence of psoriasis and depression (15.7%), as well as atopic dermatitis and depression (11.8%). These results seem plausible, since psoriasis, atopic dermatitis (about 1–3%) and depressive disorders (about 10%) are among the most common chronic/chronic-recurrent skin and mental illnesses [22]. The prevalence of atopic dermatitis is described as increasing and in childhood even affects between 10% to 30% of children [23].
In addition to genetic disposition and immunological and hormonal processes, certain medications, environmental factors, and psychosocial stressors are critical in the multifactorial etiopathogenesis of both dermatoses. On the one hand, while childhood autoimmune diseases, which include psoriasis, is discussed as caused by stressful childhood experiences [24], on the other hand, the association between atopic dermatitis and mental illness seems to be independent of the burden of serious childhood events [25]. Again, it is not surprising that the appearance and symptoms of both skin diseases can have a negative effect on mental well-being and quality of life [26,27]. Despite the prevalence of suicidal thoughts in both psoriasis patients28 and patients with atopic dermatitis [23], a recent study by Thyssen et al. [29] has shown that these patients undergo no psychiatric consultation or treatment.
Cardiovascular disease in mentally ill patients with comorbid skin disorders
Of particular note is the lifetime prevalence of 32.2% of cardiovascular diseases in psychiatric and psychosomatic patients with comorbid dermatosis. Both mental and dermatological disorders are associated with an increased risk of cardiovascular disease [30-33]. For example, Topic and colleagues 30 found somatic comorbid diseases to be significantly more common in their controlled study of 76 major depressive patients compared to the healthy control group. Cardiovascular disease was most commonly diagnosed at 46.1%, compared to 13.9% in the control group. Here, an increase in the C-reactive protein (CRP), an acute-phase protein whose synthesis is increasingly induced after the release of proinflammatory cytokines, was found to be significantly more frequent in the depressive patients than in the control subjects [30]. An elevated CRP level is again being discussed as a risk factor for atherogenesis and metabolic syndrome with an increased risk of myocardial infarction or cerebral insult [30]. In this context, it is not surprising that chronic inflammatory skin diseases are also associated with cardiovascular comorbidities, and this relationship should be of prime interest for researchers, especially in the case of patients with psoriasis [33,34]. Since fatty tissue as an endocrine organ, among other things, forms cytokines, the resulting influence on the skin’s immune system and the contribution to the pathogenesis of psoriasis has been discussed [35]. A large cohort study from Denmark with nearly 30,000 people suffering from psoriasis showed that the risk of cardiovascular disease was particularly high in psoriasis patients who also suffered from an acute depressive disorder [36].
Infectious skin diseases in mentally ill patients
Compared to the studies of Mookhoeck et al. [8] and Moftah et al. [9], which found high comorbidity rates of over 70% for infectious-parasitic dermatoses and mental disorders in primary psychiatric patients, our rate was significantly lower at 13.2%.
This is not surprising since mycoses, especially in the form of tinea manuum and tinea pedis, are very common in the general population with a lifetime prevalence of 6.5–14% [37,38] and 15–25% [39], respectively.
With regard to psychiatric patient groups, the literature suggests that patients with schizophrenia appear to be particularly susceptible to skin mycosis [40,41]. This impression could not be confirmed by our investigation, since the proportion of patients suffering from schizophrenia with dermatological comorbidities was generally low, both in the group as a whole and in the group of infectious-parasitic comorbid dermatoses. In our investigation, the strongest bacterial infectious dermatoses affected patients suffering from dementia or another organic brain disease.
This coupled with the finding that half of the patients experienced other cardiovascular risk factors or diseases underscores the importance of somatic comorbidities with consecutive immunodeficiencies as a potential pathogenetic factor for infectious dermatoses. To the best of our knowledge, up-to-date data and studies on this topic are not available, so there is a need for further research here [42]. In summary, the hypothesis that patients with a mental disorder are more likely to develop infectious-parasitic dermatoses cannot be confirmed. On the basis of our naturalistic examination of both inpatient psychiatric and inpatient psychosomatic patients, mentally ill patients do not necessarily suffer from infectious-parasitic dermatoses; however, such dermatoses occurred predominantly in patients with dementia and brain organic syndromes. Finally, our study also confirms the frequent association of psoriasis and neurodermatitis with depressive illness.
Limitations
There are some limitations of the study. In addition to the retrospective recording, the resulting small case numbers of the individual subgroups of inpatients represent a significant limiting factor with regard to general statements and can not stand for the whole population of psychiatric patients. Equally problematic is the heterogeneity of our study population, which allows only limited representative statements regarding the group of co-morbid patients, especially in the outpatient setting. It should also be taken into account that the data from the inpatients predominantly refers to seriously ill patients.
Moreover, in this retrospective and naturalistic design, not every patient may have reported a comorbid dermatological diagnosis, especially taking into account disease-specific features in dementia or schizophrenia. Furthermore, due to the computer-based search method, we cannot rule out the possibility that we failed to identify all corresponding patients. Therefore, an underestimation of the prevalence of co-morbid dermatological diseases in psychiatric and psychosomatic patients is likely. This is a strong limitation. For example, the difference found between unipolar depression and bipolar disorder found here could represent a selection bias.
Neither the severity of the mental disorder nor the dermatological disease could be validated in this study, further limiting the representativeness of our results.
Conclusions
The results indicate a link between stress-related dermatoses and mental illness, especially with regard to the clarification of further predisposing factors that contribute to the development of comorbidities in some of the patients. As such, there is a need for further research and future studies could provide important insights into these pathogenic mechanisms and the relevant therapies.
Acknowledgements
The presented partial results are data from CM’s doctoral dissertation.
Notes
The authors have no potential conflicts of interest to disclose.
Author Contributions
Conceptualization: Paraskevi Mavrogiorgou, Georg Juckel. Data curation: Corinna Mersmann, Gabriele Gerlach. Formal analysis: Corinna Mersmann, Paraskevi Mavrogiorgou, Georg Juckel. Funding acquisition: Georg Juckel. Investigation: Corinna Mersmann, Paraskevi Mavrogiorgou, Gabriele Gerlach. Methodology: Corinna Mersmann, Paraskevi Mavrogiorgou. Project administration: Paraskevi Mavrogiorgou, Georg Juckel. Resources: Stephan Herpertz, Georg Juckel. Software: Paraskevi Mavrogiorgou. Supervision: Stephan Herpertz, Georg Juckel. Validation: All authors. Visualization: Corinna Mersmann, Paraskevi Mavrogiorgou. Writing— original draft: Corinna Mersmann, Paraskevi Mavrogiorgou. Writing—review & editing: Corinna Mersmann, Paraskevi Mavrogiorgou, Georg Juckel.